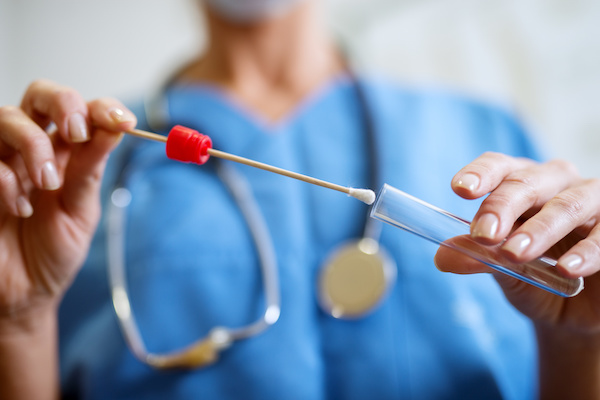
The Department of Health and Human Services’ (HHS) Substance Abuse and Mental Health Services Administration (SAMHSA) department has recently established scientific and technical guidelines for the inclusion of oral fluid specimens in the Mandatory Guidelines for Federal Workplace Drug Testing Programs using Oral Fluid (OFMG).
These guidelines will allow federal executive branch agencies to collect and test oral fluid specimen as a part of their drug testing programs.
The OFMG will be effective starting January 1st 2020. However, federal agencies will not be able to conduct oral fluid drug testing of federal employees until HHS has approved laboratories for oral fluid drug testing in accordance with the OFMG.
These guidelines may also be used by agencies such as the Department of Transportation (DOT) and Nuclear Regulatory Commission (NRC) as part of their regulated drug testing programs, but these agencies must engage in separate rule-making activities.
This is the first time since 1986 that a specimen other than urine will be able to be used in the federal agency testing programs.
Some important things to take note of with these new changes:
-The specimen collection, laboratory analysis, and medical review of oral fluid tests will mirror existing procedures and processes currently in place for urine drug testing.
-Laboratories must be approved by the HHS National Laboratory Certification Program for oral fluid drug testing. Requirements for approval will largely mirror those currently in place for urine drug testing laboratories.
-The oral fluid specimen collections must be observed, use an FDA-cleared specimen collection device, and will require split specimen procedures using 2 separate devices.
-Federal agencies will be allowed to use oral fluid testing for all of the same testing reasons as currently authorized for urine drug testing and will not be required to choose between using only urine or oral fluid.
-An approved federal custody and control form (CCF) will be used to document the oral fluid collection and establish chain of custody of the specimen.
-The drug testing panel will test for the same drugs as the federal panel for urine drug testing. Cut-off levels specific to oral fluid are included in the OFMG.
-The OFMG contains provisions for when an individual cannot provide adequate volume oral specimen or urine specimen.
-The OFMG only refers to and permits lab-based oral fluid drug testing, not instant or POCT oral fluid testing.
It has taken many years from the first time SAMHSA publicly recognized the valuable attributes of lab-based oral fluid testing until now—announcing the proposed regulations. The agency has said that their hopes with releasing oral fluid testing options will be to help reduce time and money needed in order to maintain a drug testing program, and help eliminate the ever-growing issue of drug test cheating.
To see further details on these latest updates, you can view the guideline announcement here.